MRI was performed on HIV-positive patients presenting with headaches,
altered mental status,
seizures or focal neurologic signs,
while magnetic resonance spectroscopy (MRS) was performed in patients with both neurologically subclinical disease course and with evident lesions on conventional imaging.
1. HIV-ASSOCIATED CNS ABNORMALITIES
1.1. HIV infection often results in MRI-detectable brain atrophy.
The mechanisms of HIV associated neuronal injury and loss are still unclear (opportunistic infections of the CNS,
direct HIV infection of the neuron, stimulation of cytokine production by HIV envelope glycoproteins in HIV infected neurons,
macrophages,
microglia,
and astrocytes may also injure or destroy neurons).
[5] All types of brain atrophy are more severe and more frequent in HIV-infected subjects.
[6]
1.1.1.
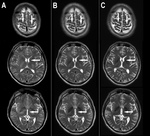
Mild atrophy for the age (Fig. 1),
associated with decreased concentration of neuronal marker,
N-acetyl aspartate,
suggesting neuronal loss/dysfunction.
1.1.2.
Marked atrophy with significantly decreased brain parenchimal volume,
most compatible with HIV encephalopathy (Fig. 2).
1.2.
AIDS dementia complex (ADC) is a clinical syndrome which comprises of a combination of cognitive,
behavioral and motor dysfunction,
attributed to direct effect of HIV on brain,
in absence of opportunistic infection. This is most frequent neurological manifestation of HIV infection.
The natural history is progression to a global dementia (Fig. 3).
The associated imaging appearance is generally referred to as AIDS encephalopathy,
HIV encephalopathy or HIV encephalitis (Fig. 2).
Before HAART (highly active antiretroviral therapy) was available,
the median survival time of an AIDS dementia complex patient after the onset of dementia was 6 months.
Improvement in survival was noted in patients with AIDS dementia complex treated with HAART. [7]
MRI findings are non-specific and include symmetrical cerebral atrophy with symmetric periventricular and deep white matter T2 hyperintensity (confluent or patchy) with no mass effect and no enhancement.
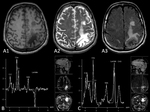
Investigators using 1H MRS have reported decreased N-acetyl aspartate (NAA) and increased choline (Cho) in cases of AIDS with low CD4 lymphocyte counts,
neurologic evidence for ADC,
and atrophy.
In patients who are cognitively normal and clinically asymptomatic,
1H MRS in the subcortical region demonstrates elevated Cho concentrations early in the disease before symptoms,
whereas NAA is abnormally low only with severe neuropsychologic impairments.
[8] Severity is related to the patient's viral load,
and can regress with HAART.
[9] (Fig. 4)
2. OPPORTUNISTIC INFECTIONS
The advent of HAART,
which has been used in Western countries to treat HIV-infected patients since 1996,
has resulted in a decline in the incidence of neurologic complications,
especially those caused by opportunistic infections.
[10,11]
Although CNS opportunistic infections are decreasing in frequency,
they continue to have a devastating effect on HIV-positive individuals,
especially those in whom diagnosis is delayed or HIV treatment is inadequate.
Mortality is often high even with appropriate treatment,
and recurrences and residual neurological deficits are common.
[12]
2.1.
Progressive multifocal leukoencephalopathy (PML) (Fig. 5)
PML is a rare but severe CNS opportunistic infection that is caused by the reactivation of latent JC virus (John Cunningham virus,
which is a DNA papovavirus) in immunosuppressed individuals.
The incidence of PML has declined from seven cases per 1000 patient-years before the introduction of combination antiretroviral therapy (cART) to 0,7 per 1000.
[13]
Clinically,
PML may present with nonspecific headache,
visual disturbance,
dementia,
hemiparesis,
cognitive impairment or seizure (as PML can also involve the grey matter).
[14]
Classic PML presents as a subacute illness without constitutional symptoms in patients with severe immunodeficiency.
Progressive focal neurology,
mainly motor deficit,
altered mental or mood status,
ataxia or cortical visual symptoms,
develop over weeks to months.
Seizures may rarely occur.
[15]
The presence of the focal features helps distinguish the cognitive syndrome associated with PML from HIV encephalopathy.
On MR images,
there are typically multifocal,
asymmetric areas of hyperintensity in the periventricular and subcortical white matter.
These lesions are frequently bilateral and multiple,
although they may occasionally be solitary.
Subcortical U-fiber involvement is frequently seen,
a finding that provides a sharp contrast with the overlying gray matter.
Mass effect and hemorrhage are unusual,
but,
if present,
they are typically mild.
Typically,
PML lesions do not enhance,
but faint peripheral enhancement has been described.
Evaluation of diffusion-weighted imaging in patients with PML has been limited.
MR spectroscopy demonstrates a reduction in NAA,
presence of lactate,
and increased amounts of choline and lipids.
[16]
A definitive diagnosis of PML is established by the detection of JC virus in CSF by PCR.
Brain biopsy might occasionally be required to confirm the diagnosis.
[12]
2.2.
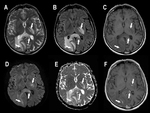
Cytomegalovirus (CMV) infection (Fig. 6)
The ubiquitous herpes virus,
cytomegalovirus is endemic worldwide and usually causes asymptomatic or clinically benign infections.
With clinical progression of HIV,
latent CMV reactivates,
leading to viraemia and,
in a proportion,
end-organ disease.
CNS involvement typically assumes the form of meningoencephalitis or ventriculitis,
but it can also take the form of myelitis,
polyradiculitis,
and retinitis [16].
Patients with cytomegalovirus encephalitis commonly present with rapidly progressive encephalopathy [12].
Focal neurological signs and cranial nerve palsies are uncommon to rare.
Atrophy,
periventricular or subependymal enhancement,
and periventricular high intensity on T2WI/FLAIR may be associated with CMV infection but can also be seen in the plethora of other diseases affecting the brain in AIDS.
[8]
PCR of CSF is highly sensitive and specific in the diagnosis of CMV encephalitis.
A neutrophil predominant CSF pleocytosis with negative bacterial culture is highly suggestive of cytomegalovirus infection.
[12]
2.3.
Toxoplasmosis (Fig. 7)
Cerebral toxoplasmosis,
the most common opportunistic CNS infection in AIDS patients,
occurs in 15%–50% of cases.
[17] With the introduction of HAART,
the number of cases has declined.
[18]
Toxoplasmosis typically results in necrotizing encephalitis.
The regions of the corticomedullary junction,
basal ganglia,
and thalamus are affected most often; however,
the brainstem may also be involved.
Lesions from toxoplasmosis are usually multiple,
and only in approximately 14% of cases are they solitary.
Lesions are typically hypo- to isointense on T2WI and are surrounded by high-signal-intensity vasogenic edema.
Hemorrhage may be seen occasionally,
a finding that can help differentiate toxoplasmosis from lymphoma,
which typically does not hemorrhage before treatment.
Postcontrast MR imaging reveals multiple nodular lesions or ring-enhancing lesions.
Occasionally,
a small eccentric nodule rests alongside an enhancing ring: the “target sign”.
This finding is highly suggestive of toxoplasmosis; however,
it is relatively insensitive and is seen in less than 30% of cases.
Toxoplasmosis occasionally involves the corpus callosum and may mimic a glioblastoma multiforme.
[16]
2.4.
Unusual infections
2.4.1.
Bacterial Infections
Pyogenic intracranial infections (bacterial meningitis,
pyogenic abscesses (Fig. 8)) are relatively uncommon in the AIDS population.
AIDS patients are subject to the same types of bacterial infections seen in immunocompetent patients,
and Staphylococcus and Streptococcus are the most common causative organisms.
The imaging findings seen in AIDS patients are identical to those seen in immunocompetent patients.
2.4.2.
CNS tuberculosis ( Fig. 9 )
CNS tuberculosis can result from reactivation of a previous infection or from a primary,
newly acquired infection.
The predominant mechanism of disease spread is hematogenous.
Mortality from HIV-associated tuberculous meningitis often exceeds 50%,
which is roughly double the rate in patients without HIV.
[19] Even with the addition of cART,
mortality is not improved.
[20]
The most common intracranial manifestation of tuberculosis is meningitis,
which is usually more prominent in the basilar cisterns,
especially around the circle of Willis.
[16] Tuberculous meningitis usually presents as T2/FLAIR hyperintense,
thick or nodular meningeal enhancement in the basal cisterns.
Communicating hydrocephalus,
which is a common sequela of tuberculous meningitis,
primarily results from obstruction of the basal cisterns by the dense inflammatory exudates.
Occasionally,
the hydrocephalus may be attributable to a focal parenchymal lesion with mass effect or entrapment of a ventricle by granulomatous ependymitis.
[3]
Tuberculomas are granulomas that result either from hematogenous spread or extension from cerebrospinal fluid infection via cortical veins or small penetrating arteries (84).
The majority of tuberculomas are supratentorial and may be solitary or multiple; however,
they can also be found in subdural,
epidural,
and subarachnoid spaces (Fig 19) (84).
Tuberculomas are hypointense on T2-weighted MR images in the early stages; as they mature,
they develop a hypointense center surrounded by an isointense capsule,
which corresponds to solid caseation necrosis.
They may further progress to abscess formation with a hyperintense center.
However,
some tuberculomas have a hyperintense center without abscess formation,
an appearance that makes them difficult to distinguish from lesions of toxoplasmosis or lymphoma.
On postcontrast images,
noncaseating tuberculomas demonstrate nodular homogeneous enhancement.
Caseating tuberculomas have ring enhancement.
[16]
Tuberculous abscesses are rare,
but more common in HIV-infected patients than in non HIV-infected patients.
Among patients with CNS tuberculosis,
4%–8% of those without HIV infection developed abscesses,
compared with up to 20% in one group of HIV patients.
[21]
Abscesses tend to be larger (frequently greater than 3 cm) than tuberculomas,
which are typically less than 1 cm,
and have a more accelerated clinical course.
They are mostly multiloculated,
and central liquefaction with pus formation appears hyperintense on T2W images.
Abscesses are also more frequently solitary than tuberculomas.
Significant edema and mass effect favor abscess.
Similar to other abscesses,
tuberculous abscesses demonstrate ring enhancement after administration of contrast material,
which can pose problems in differentiation from abscesses of other etiologies,
especially in the absence of associated findings such as basal meningitis,
hydrocephalus and basal ganglia infarcts.
Infarction is another common complication of CNS tuberculosis,
a result of spasm and thrombosis as arterial vessels course through the gelatinous basal exudates.
The most common locations are the regions of basal ganglia.
[3]
Imaging does not necessarily show tuberculoma in cases of tuberculous meningitis,
and obviously not all patients with tuberculoma have meningeal enhancement.
[8]
2.4.3.
Fungal infections
Most fungal infections of the CNS in immunocompromised patients,
results from hematogenous spread from a pulmonary focus,
or the fungus may also directly invade the brain via the sinus.
The most commonly encountered fungi within this population is Cryptococcus.
Other less common fungal infections affecting HIV-positive patients include those caused by Aspergillus,
Candida and Histoplasma.
[8,16]
2.4.3.1.
Cryptococcosis
Cryptococcosis most likely spreads to the CNS by means of hematogenous dissemination from a pulmonary focus; however,
reactivation of a latent cryptococcal infection is also possible.
[16]
CNS cryptococcosis develops in up to 11% of patients with AIDS.
MR may be normal,
or can demonstrate a spectrum of abnormalities.
Recognition of dilated Virchow Robin spaces in young immunosuppressed patients should raise a red flag concerning the possibility of intracranial Cryptococcus.
These dilated spaces filled with gelatinous cysts,
in and adjacent to the basal ganglia and the corticomedullary junction,
may or may not enhance.
Another important pattern is that of multiple miliary enhancing parenchymal and leptomeningeal nodules with involvement of the choroid plexus in the trigone as well as the spinal cord and spinal nerve roots.
Less common findings include widening of the basal cisterns,
hydrocephalus,
and diffuse atrophy.
[8]
2.4.3.2.
Aspergillosis
Aspergillosis involves the brain in an aggressive form,
producing meningitis and meningoencephalitis with subsequent hemorrhagic infarction.
Less malignant presentations include solitary cerebral abscess or isolated granulomas.
Magnetic resonance shows high-intensity lesions on T2WI/ FLAIR and at times on T1WI.
If the site of origin is the paranasal sinus,
one may see decreased intensity on T2WI/FLAIR secondary to calcification or manganese accumulation.
There may be evidence of infarction with or without enhancement.
The MR findings are nonspecific but may cause curiosity when the lesions do not enhance,
as occurs sometimes.
This may be from the invasion of blood vessels restricting delivery of the contrast to the site.
Basal ganglia lesions are common with fungi and they,
like tuberculosis,
can have a crenated appearance.
[8]
2.4.4.
Neurosyphilis
HIV-infected patients are more likely to progress to clinical neurosyphilis,
and neurologic symptoms develop after shorter latent periods than in non-AIDS population.
[22]
Neurosyphilis has a wide variety of imaging findings.
Mild to moderate atrophy,
white matter lesions,
cortical and subcortical infarctions,
gummas,
leptomeningeal enhancement,
and arteritis have all been reported.
Typical imaging findings are ischemic infarcts in the perforator vessel territory of the basal ganglia or brainstem or in the large vessel territory of the middle cerebral artery.
Syphilitic cerebral gummas are uncommon.
They appear as isolated,
peripherally located nodules isointense to gray matter on T1W sequences,
usually enhance homogeneously,
and are hyperintense on T2W sequences.
Cranial nerve involvement,
particularly of the optic and vestibulocochlear nerves,
has also been reported.
[3,16]
3.
CNS NEOPLASMS
3.1.
CNS lymphoma
HIV infection is a well-established risk factor for primary CNS lymphoma.
Since HAART/cART was introduced,
the incidence among people with HIV infection has substantially declined and it is now a rare disease.
[12]
Primary CNS lymphoma (Fig. 10) includes single or multiple areas of involvement with mass effect without a great deal of edema relative to the tumor size.
There is a marked predilection for the basal ganglia,
cerebellar hemispheres,
thalamus,
brain stem,
corpus callosum,
and subependymal region.
On MR,
these lesions may be isointense or of low intensity on T1WI and variable intensity on T2WI/FLAIR.
Most lesions enhance in a solid or ring fashion,
although nonenhancing lymphoma has been reported.
DWI often shows restricted diffusion.
[8]
Secondary CNS lymphoma is typically a non-Hodgkin lymphoma,
and by definition has systemic disease at the time of presentation,
with secondary involvement of the central nervous system.
Unlike primary CNS lymphoma it more commonly involves the leptomeninges and is uncommonly detectable on CT/MR,
with malignant cells found of CSF aspiration. (Table 1,
Table 2) [23]
4.
DRUG-RELATED COMPLICATIONS OF THERAPY
The introduction of highly active antiretroviral therapy (HAART) / combination antiretroviral therapy (cART),
has led to a significant reduction in AIDS-related morbidity and mortality.
[24]
Antiretroviral therapy can have a wide range of adverse effects on the human body,
including drug interactions,
toxic effects and IRIS.
4.1. Immune reconstitution inflammatory syndrome (IRIS) (Fig. 6C)
IRIS is an abnormal inflammatory reaction to a subclinical or previously treated infection that occurs after initiation of HAART,
despite a decreasing viral load and a rising CD4+ T-cell count,
and it typically occurs within 60 days after the start of HAART.
[25] The phenomenon was initially recognized in the mid-1990s when the clinical condition of some patients deteriorated after HAART was started.
There are two common IRIS scenarios.
The first is the “unmasking” of an occult opportunistic infection.
The second is the “paradoxical” symptomatic relapse of a prior infection despite microbiologic treatment success.
Often in paradoxical IRIS,
microbiologic cultures are sterile.
In either scenario,
there is hypothesized reconstitution of antigen-specific T cell-mediated immunity with activation of the immune system following HIV therapy against persisting antigen,
whether present as intact organisms,
dead organisms,
or debris.
[26]
Occasionally,
IRIS may be associated with a fatal outcome.
[27]
The neuroimaging findings vary,
depending on the underlying pathologic conditions,
or may be atypical.